Recombinant viral vectors, in particular those based on adeno-associated viruses (AAVs), have proven incredibly useful for studying the complexities of the brain. They make it possible to test hypotheses about gene and cell function by expressing, knocking down, or editing genes of interest in vivo and by monitoring or modulating the state of specific cell populations using a rapidly expanding collection of genetically encoded sensors and actuators. In addition, AAV-based gene therapies are under development for numerous diseases, including many in the CNS.
The vector engineering team within the Stanley Center for Psychiatric Research is focused on using high-throughput protein engineering and library selection methods to develop AAVs with enhanced capabilities, such as the ability to provide efficient CNS-wide transduction via the vasculature or expression that is restricted to specific cell types.
AddGene: https://www.addgene.org/Ben_Deverman/
AAV Capsid Engineering
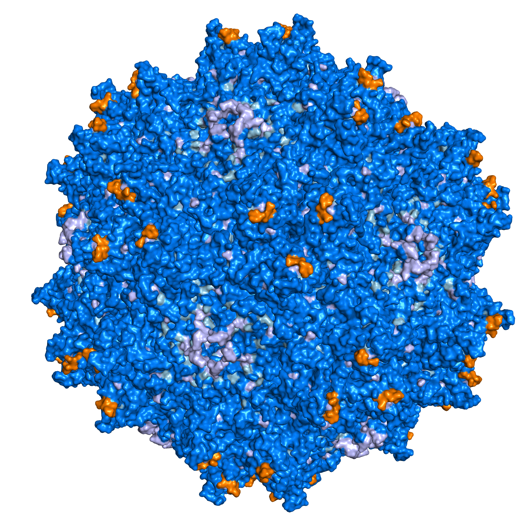
The AAV capsid is the protein shell of the virus that protects the recombinant genome and determines the tropism of the virus. Over a hundred natural AAV isolates have been identified; however, many of these AAVs exhibit broad, overlapping tropisms with insufficient expressivity. Therefore, we are interested in evolving capsids with specific tropisms that extend beyond what nature has provided using protein engineering. We recently described a novel in vivo cell type–specific capsid selection method called CREATE (Cre Recombinase-based AAV Targeted Evolution). We used this selection method to identify numerous capsids that more efficiently transduce cells throughout the central and peripheral nervous system in the adult mouse following noninvasive intravascular delivery. Most notably we developed several capsids, including AAV-PHP.B and AAV-PHP.eB, that efficiently transduce the mouse CNS following passage across the blood-brain barrier (Deverman et al., Nature Biotechnology 2016 and Chan et al., Nature Neurosci 2017), and AAV-PHP.S, which efficiently transduces neurons throughout the peripheral nervous system. We continue to use CREATE and other high-throughput selection and screening methods to develop capsids for additional applications.
Gene Expression and Regulation

In addition to controlling AAV tropism, we also develop customized AAV genomes that provide gene expression in defined cell populations without the use of transgenics. We customize the recombinant genome with transcriptional, post-transcriptional and translational control elements to develop expression systems that are restricted to specific cell types, circuits and cellular states.